
| Soluble VEGF R1/sFlt-1 | |
| Biomarker for Cardiovascular Disease and Tumor | |
Alternative name:
|
|
|
|
|
| Vascular endothelial growth factor-B acts as a coronary growth factor in transgenic rats without inducing angiogenesis, vascular leak, or inflammation | |
| Background- Vascular endothelial growth factor-B (VEGF-B) binds to VEGF receptor-1 and neuropilin-1 and is abundantly expressed in the heart, skeletal muscle, and brown fat. The biological function of VEGF-B is incompletely understood. Methods and Results- Unlike placenta growth factor, which binds to the same receptors, adeno-associated viral delivery of VEGF-B to mouse skeletal or heart muscle induced very little angiogenesis, vascular permeability, or inflammation. As previously reported for the VEGF-B(167) isoform, transgenic mice and rats expressing both isoforms of VEGF-B in the myocardium developed cardiac hypertrophy yet maintained systolic function. Deletion of the VEGF receptor-1 tyrosine kinase domain or the arterial endothelial Bmx tyrosine kinase inhibited hypertrophy, whereas loss of VEGF-B interaction with neuropilin-1 had no effect. Surprisingly, in rats, the heart-specific VEGF-B transgene induced impressive growth of the epicardial coronary vessels and their branches, with large arteries also seen deep inside the subendocardial myocardium. However, VEGF-B, unlike other VEGF family members, did not induce significant capillary angiogenesis, increased permeability, or inflammatory cell recruitment. Conclusions- VEGF-B appears to be a coronary growth factor in rats but not in mice. The signals for the VEGF-B-induced cardiac hypertrophy are mediated at least in part via the endothelium. Because cardiomyocyte damage in myocardial ischemia begins in the subendocardial myocardium, the VEGF-B-induced increased arterial supply to this area could have therapeutic potential in ischemic heart disease. | |
| Bry M, et al. .Circulation. 2010 Oct 26;122(17):1725-33. Epub 2010 Oct 11. | |
| Up-regulation of Soluble Vascular Endothelial Growth Factor Receptor-1 Prevents Angiogenesis in Hypertrophied Myocardium | |
| Aims Inadequate capillary growth in pressure-overload hypertrophy impairs myocardial perfusion and substrate delivery, contributing to progression to failure. Capillary growth is tightly regulated by angiogenesis growth factors like vascular endothelial growth factor (VEGF) and endogenous inhibitors such as the splice variant of VEGF receptor-1, sVEGFR-1. We hypothesized that inadequate expression of VEGF and up-regulation of VEGFR-1 and its soluble splice variant, sVEGFR-1, restrict capillary growth in pressure-overload hypertrophy. Methods Neonatal New Zealand White rabbits underwent aortic banding. mRNA (qRT-PCR) and protein levels (immunoblotting) were determined in hypertrophied and control myocardium (7/group) for total VEGF, VEGFR-1, sVEGFR-1, VEGFR-2 and phospho-VEGFR-1and -R-2. Free VEGF was determined by ELISA in hypertrophied myocardium, controls and hypertrophied hearts following inhibition of sVEGFR-1 with Placental Growth Factor (PlGF). Results VEGFR-1 and sVEGFR-1 mRNA (7-fold up-regulation, p=0.001) and protein levels were significantly up-regulated in hypertrophied hearts versus controls (VEGFR-1: 44±8 vs. 23±1, p=0.031; sVEGFR-1: 71±13 vs. 31±3, p=0.016). There was no change in VEGF and VEGFR-2 mRNA or protein levels in hypertrophied compared to controls hearts. A significant decline in free, unbound VEGF was found in hypertrophied myocardium which was reversed following inhibition of sVEGFR-1 with PlGF, which was accompanied by phosphorylation of VEGFR-1 and VEGFR-2. Conclusions Up-regulation of the soluble VEGFR-1 in pressure-loaded myocardium prevents capillary growth by trapping VEGF. Inhibition of sVEGFR-1 released sufficient VEGF to induce angiogenesis and preserved contractile function. These data suggest sVEGFR-1 as possible therapeutic targets to prevent heart failure. | |
| Kaza E,, et al. Cardiovasc Res. 2010 Oct 8. [Epub ahead of print] | |
| Biological variations in plasma VEGF and VEGFR-1 may compromise their biomarker value in colorectal cancer | |
INTRODUCTION: Vascular Endothelial Growth Factor (VEGF) plays a prominent role in tumor angiogenesis and plasma VEGF concentration may carry prognostic information in colorectal cancer. The VEGF receptor 1 (VEGFR-1) is a regulatory receptor which is shredded into plasma of patients with colorectal cancer. For both molecules, large biological variation and lack of standardization of assay procedures are major challenges. METHODS: We investigated pre-analytical, analytical, as well as short term and long term biological variation of plasma VEGF and VEGFR-1 in volunteers. In addition, we evaluated plasma VEGF and VEGFR-1 as markers of colorectal disease in a case-control study on four groups of 77 individuals undergoing bowel endoscopy. Groups were categorized as 'no findings', 'non-malignant findings', 'adenoma', or 'colorectal cancer'. RESULTS: In the studies on variation, temperature and delay before centrifugation significantly influenced plasma VEGF and, to a minor extent, plasma VEGFR-1 concentrations. In addition, we found large biological variations with CV up to 69.2% for VEGF and CV up to 35.9% for VEGFR-1. For both molecules the intra-subject variation exceeded the inter-subject variation. In the case control study neither plasma VEGF nor VEGFR-1 was able to differentiate between the four groups of individuals although plasma VEGFR-1 was significantly lower in patients with 'no findings'. CONCLUSION: There was no difference in plasma VEGF or VEGFR-1 between patients with no findings, benign disease, pre-malignant findings, and malignant findings after endoscopy. The poor discrimination between patients may be explained by the large inter- and intra-subject variations found for both molecules in volunteers. |
|
Svendsen MN,, et al. Scand J Clin Lab Invest. 2010 Nov;70(7):503-11. Epub 2010 Sep 27. |
|
| Effect of intensive insulin therapy on macular biometrics, plasma VEGF and its soluble receptor in newly diagnosed diabetic patients | |
BACKGROUND: To evaluate whether intensive insulin therapy leads to changes in macular biometrics (volume and thickness) in newly diagnosed diabetic patients with acute hyperglycaemia and its relationship with serum levels of vascular endothelial growth factor (VEGF) and its soluble receptor (sFlt-1). METHODS: Twenty-six newly diagnosed diabetic patients admitted to our hospital to initiate intensive insulin treatment were prospectively recruited. Examinations were performed on admission (day 1) and during follow-up (days 3, 10 and 21) and included a questionnaire regarding the presence of blurred vision, standardized refraction measurements and optical coherence tomography. Plasma VEGF and sFlt-1 were assessed by ELISA at baseline and during follow-up. RESULTS: At study entry seven patients (26.9%) complained of blurred vision and five (19.2%) developed burred vision during follow-up. Macular volume and thickness increased significantly (p = 0.008 and p = 0.04, respectively) in the group with blurred vision at day 3 and returned to the baseline value at 10 days. This pattern was present in 18 out of the 24 eyes from patients with blurred vision. By contrast, macular biometrics remained unchanged in the group without blurred vision. We did not detect any significant changes in VEGF levels during follow-up. By contrast, a significant reduction of sFlt-1 was observed in those patients with blurred vision at day 3 (p = 0.03) with normalization by day 10. CONCLUSION: Diabetic patients with blurred vision after starting insulin therapy present a significant transient increase in macular biometrics which is associated with a decrease in circulating sFlt-1. |
|
Hernández C, et al. Diabetes Metab Res Rev. 2010 Jul;26(5):386-92. |
|
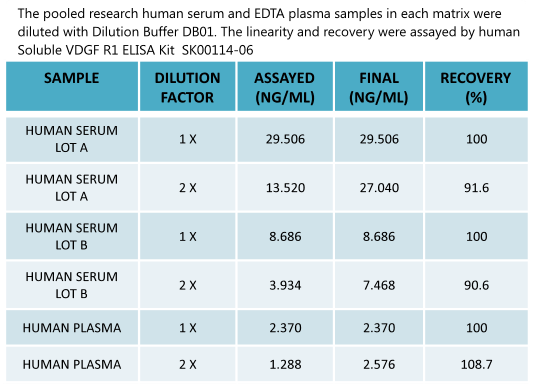
New Human Soluble VEGF -RI ELISA Kit SK00114-06 enables to detect circulating samples |
|
 |
|
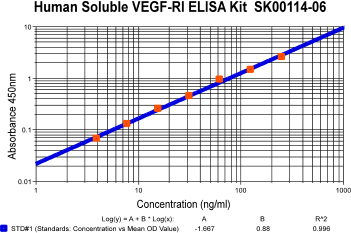 |
Human Soluble VEGFR1/sFlt-1 ELISA Code No.: SK00114-06 Size: 96 T Price: $360.00 USD Standard Range:3.9-250 ng/ml Dynamic Range: 3.9-250 ng/ml Sensitivity:100 pg/ml Sample Type: serum, EDTA plasma Sample requres: 250 ul IntraCV: 4-6% InterCV: 8-10% Protocol: PDF |
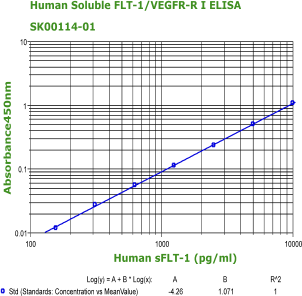 |
Human Soluble VEGFR1/sFlt-1 ELISA Code No.: SK00114-01 Size: 96 T Price: $360.00 USD Standard Range:156-10000 pg/ml Dynamic Range: 156-10000 pg/ml Sensitivity:78 pg/ml Sample Type: cell cultures, serum, plasma Sample requres: 250 ul IntraCV: 4-6% InterCV: 8-10% Protocol: PDF |
 |
Human Soluble VEGFR1/sFlt-1
(32-327) ELISA Code No.: SK00114-09 Size: 96 T Price: $460.00 USD Standard Range:3.9-250 ng/ml Sensitivity:260 pg/ml Sample Type: cell cultures, serum, plasma Sample requres: 250 ul IntraCV: 4-6% InterCV: 8-10% Protocol: PDF |
|
Human
VEGF-R1/sFlt-1 ECD (32-327)Recombinant Code No.: 00114-01-100 Size: 100 ug Price: $360.00 USD Protein ID:P17948 Gene ID: 2321 MW:35 KD Tag: His Tag on N-Terminus Expressed: E. Coli Purity: 95% Data Sheet: PDF |
| Human
VEGF-R1/sFlt-1 ECD (335-654)Recombinant Code No.: 00114-06-100 Size: 100 ug Price: $360.00 USD Protein ID:P17948 Gene ID: 2321 MW:54 KD Tag: His Tag on N-Terminus Expressed: E. Coli Purity: 95% Data Sheet: PDF |
|
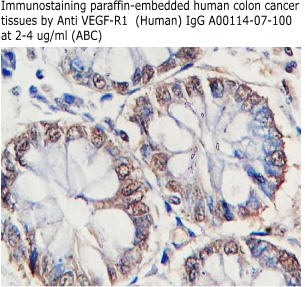 |
Anti Human
sVEGF-R1/sFlt-1 IgG Code No.: A00114-07-100 Size: 100 ug Price: $220.00 USD Host: Rabbit Antigen: human sVEGFR-1/sFlt-1 (32-327) Rec. Ab Type: Polyclonal IgG Purification: Protein A Applications: E, WB Working Dilution: 2 ug/ml) Data Sheet: PDF |
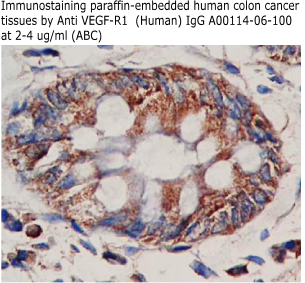 |
Anti Human
sVEGFR-1/sFlt-1 IgG Code No.: A00114-06-100 Size: 100 ug Price: $220.00 USD Host: Rabbit Antigen: human sVEGFR-1/sFlt-1 (335-654) Rec. Ab Type: Polyclonal IgG Purification: Protein A Applications: E,WB Working Dilution: 2 ug/ml) Data Sheet: PDF |
|
|
| Name | Code No. |
Size |
Price ($) |
| Soluble VEGF-R1/sFlt-1 (Human) ELISA Kit (for serum or plasma sample assay) | SK00114-06 | 96 T | 360.00 |
| Soluble VEGF-R1/sFlt-1 (32-327) (Human) ELISA Kit | SK00114-09 | 96 T | 460.00 |
| Soluble VEGF-R1/sFlt-1 (Human) ELISA Kit | 96 T |
360.00 |
|
| Soluble VEGF-R1/sFlt-1 (Mouse) ELISA Kit | 96 T |
360.00 |
|
| sVEGFR1/sFlt-1 (Human) Rec. (human cells) | 10 ug |
100.00 |
|
| sVEGFR1/sFlt-1 (Human) Rec. (human cells) | 50 ug |
inquire |
|
| sVEGFR1/sFlt-1 (32-327) (Human) Rec. | 100 ug |
360.00 |
|
| sVEGFR1/sFlt-1 (335-654) (Human) Rec. | 100 ug |
360.00 |
|
| Anti sVEGFR1/sFlt-1(Human) IgG | 100 ug |
220.00 |
|
| Anti sVEGFR1/sFlt-1(Human) IgG | 100 ug |
220.00 |
|
| Anti sVEGF-R1/sFlt-1 (Human) Antibody | 100 ul |
360.00 |
|
| Anti sVEGF-R1/sFlt-1 (Human) Monoclonal Antibody | 100 ug |
360.00 |
|
| Anti sVEGF-R1/sFlt-1 (32-327) (Human) Monoclonal Antibody | 100 ug |
260.00 |
|
| Anti sVEGF-R1/sFlt-1 (32-327) (Human) Monoclonal Antibody | 100 ug |
260.00 |
|
| Anti sVEGF-R1/sFlt-1 (335-654) (Human) Monoclonal Antibody | 100 ug |
260.00 |
|
| Anti sVEGF-R1/sFlt-1 (335-654) (Human) Monoclonal Antibody | 100 ug |
260.00 |
|